 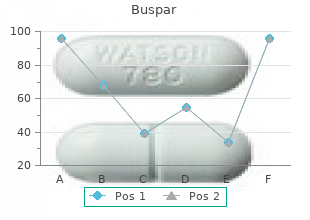
Thus, dose increases and repeated dosing may lead to somewhat higher blood levels of unchanged buspirone than would be predicted from results of single-dose studies. This suggests that food may decrease the extent of presystemic clearance of buspirone (seeĪ multiple-dose study conducted in 15 subjects suggests that buspirone has nonlinear pharmacokinetics. Max) of unchanged buspirone increased by 84% and 116%, respectively, but the total amount of buspirone immunoreactive material did not change. They were given a 20 mg dose with and without food the area under the plasma concentration-time curve (AUC) and peak plasma concentration (C The effects of food upon the bioavailability of buspirone hydrochloride tablets have been studied in eight subjects. The single-dose bioavailability of unchanged buspirone when taken as a tablet is on the average about 90% of an equivalent dose of solution, but there is large variability. Peak plasma levels of 1 ng/mL to 6 ng/mL have been observed 40 to 90 minutes after single oral doses of 20 mg. Following oral administration, plasma concentrations of unchanged buspirone are very low and variable between subjects. In a radiolabeled study, unchanged buspirone in the plasma accounted for only about 1% of the radioactivity in the plasma. Some studies do suggest that buspirone may have indirect effects on other neurotransmitter systems.īuspirone hydrochloride tablets are rapidly absorbed in man and undergoes extensive first-pass metabolism. In vivo when tested in preclinical models.īuspirone has moderate affinity for brain DĢ-dopamine receptors. Buspirone has no significant affinity for benzodiazepine receptors and does not affect GABA binding In vitro preclinical studies have shown that buspirone has a high affinity for serotonin (5-HTġA) receptors. It also lacks the prominent sedative effect that is associated with more typical anxiolytics. Buspirone differs from typical benzodiazepine anxiolytics in that it does not exert anticonvulsant or muscle relaxant effects. The mechanism of action of buspirone is unknown. Buspirone hydrochloride tablets contain the following inactive ingredients: colloidal silicon dioxide, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and croscarmellose sodium. A single 30 mg tablet can provide the following doses: 30 mg (entire tablet), 20 mg (two thirds of a tablet), 15 mg (one half of a tablet), or 10 mg (one third of a tablet). Thus, a single 15 mg tablet can provide the following doses: 15 mg (entire tablet), 10 mg (two thirds of a tablet), 7.5 mg (one half of a tablet), or 5 mg (one third of a tablet). These tablets are scored so they can be either bisected or trisected. 
The 15 mg and 30 mg tablets are provided in multi-scored tablet design. 
Thus, the 5 mg tablet can also provide a 2.5 mg dose, and the 10 mg tablet can provide a 5 mg dose. The 5 mg and 10 mg tablets are scored so they can be bisected. Buspirone hydrochloride tablets are supplied as tablets for oral administration containing 5 mg, 7.5 mg, 10 mg, 15 mg, or 30 mg of buspirone hydrochloride, USP (equivalent to 4.6 mg, 6.9 mg, 9.1 mg, 13.7 mg, and 27.4 mg of buspirone free base, respectively). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
